Chemical Engineering Archive: Questions from December 09, 2022
-
Note: Remember for reversible processes ΔS=Q/T (Do not forget the sign of the heat. if it enters or leaves the reserve) A completely reversible heat pump produces heat at a rate of 3.0 MW to heat a s
b. Entropía: \[ \Delta S=\frac{Q_{v v}}{T} \quad T \Delta S=\Delta U+P \Delta V \quad T \Delta S=\Delta h-V \Delta P \quad Q_{v v}=T \Delta S \] Gases idelaes@ \( \mathrm{C}_{\mathrm{p}} \mathrm{y} \1 answer -
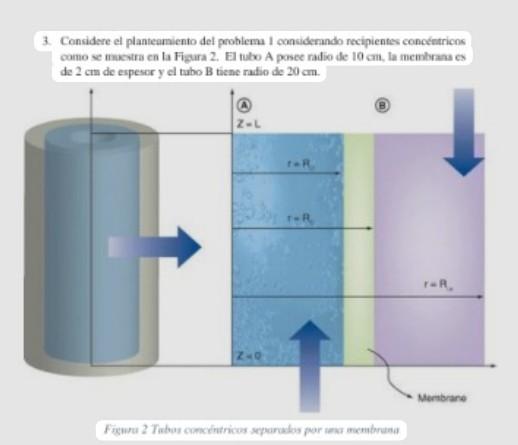
3. Considere el planteamiento del problema I considerando recipientes conceintricns como se inkevtra cn la Figura 2. El tulo A posee radio de \( 10 \mathrm{~cm} \). la memlorna es0 answers


