¡Tu solución está lista!
Nuestra ayuda de expertos desglosó tu problema en una solución confiable y fácil de entender.
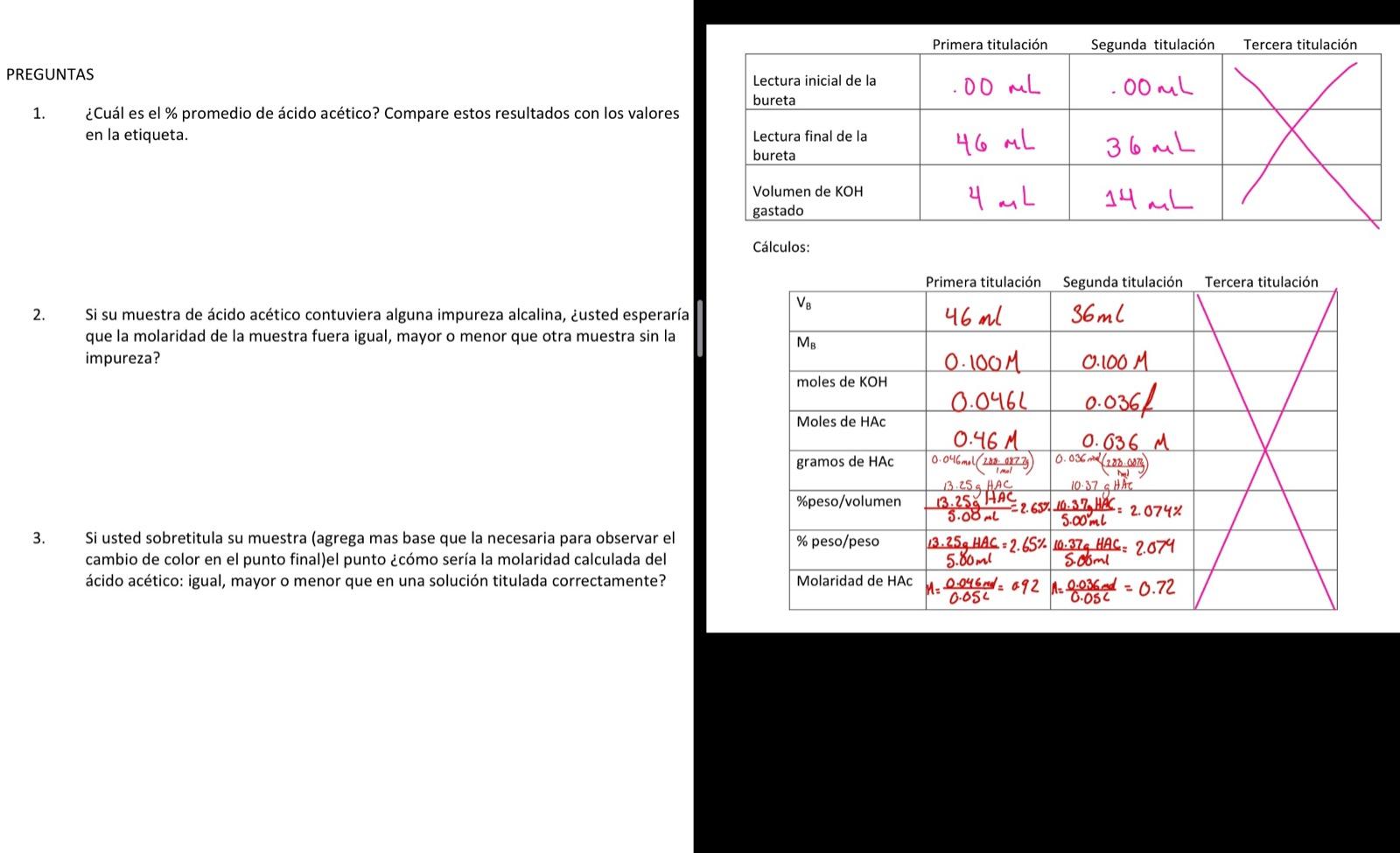
Mira la respuestaMira la respuesta done loadingPregunta: traducion 1. What is the average % of acetic acid? Compare these results with the values In the label. (4.5% in weight) 2. If your sample of acetic acid contained some alkaline impurity, would you expect the molarity of the
traducion
1. What is the average % of acetic acid? Compare these results with the values In the label. (4.5% in weight)
2. If your sample of acetic acid contained some alkaline impurity, would you expect the molarity of the sample to be the same, greater, or less than another sample without the impurity?
3. If you overtitrate your sample, what would the calculated molarity of acetic acid look like: equal to, greater than, or less than in a correctly titrated solution?
- Esta es la mejor manera de resolver el problema.Solución
1)percnetage/volume acetic acid in the 1st two trial is = 2.65% and 2.074% So the average percnetage/volume of acetic acid will be (2.65+2.074)/2 %= 2.347 % In the label the average…
Mira la respuesta completa
Estudia mejor, ¡ahora en español!
Entiende todos los problemas con explicaciones al instante y pasos fáciles de aprender de la mano de expertos reales.