¡Tu solución está lista!
Nuestra ayuda de expertos desglosó tu problema en una solución confiable y fácil de entender.
Mira la respuestaMira la respuesta done loadingPregunta: 1) Write the balanced chemical equation for the reaction carried out. data and results 1. volume of commercial H2O2 solution used =5.00 mL 2. Mass of commercial H2O2 solution 3. Reading of the water level in
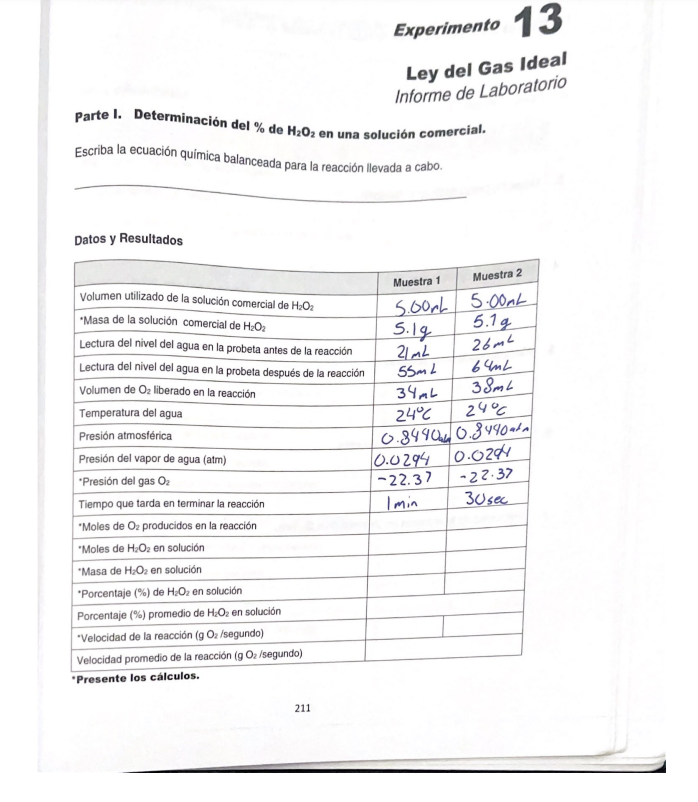
1) Write the balanced chemical equation for the reaction carried out.
data and results
1. volume of commercial H2O2 solution used =5.00 mL
2. Mass of commercial H2O2 solution
3. Reading of the water level in the cylinder before the reaction
4. Reading of the water level in the cylinder after the reaction
5. Volume of O2 released in the reaction
6. Water temperature
7. Atmospheric pressure
8. water vapor pressure atm
9. O2 gas pressure
10. Time it takes for the reaction to complete
11. Moles of O2 produced in the reaction
12. Moles of H2O2 in solution
13. Mass of H2O2 in solution
14. % of H2O2 in solution
15. % de average de H2O2 in solution
16. Reaction rate (g O2/second)
17. Average reaction rate (g O2/second)- Esta es la mejor manera de resolver el problema.Solución
Reaction: 2H2O2→2H2O+O2 Explanation: This reaction is basically the spon…
Mira la respuesta completa
Estudia mejor, ¡ahora en español!
Entiende todos los problemas con explicaciones al instante y pasos fáciles de aprender de la mano de expertos reales.